Increasing numbers of people are seeking cosmetic procedures to reduce the appearance of wrinkles, crow’s feet, and other signs of ageing.
Face and hand wrinkles and volume loss can be reduced by injecting dermal fillers into the areas affected by ageing or certain medical conditions. In studies of FDA-approved dermal fillers, patients generally report satisfaction with their treatment outcomes.
Fillers in the skin aren’t right for everyone. People with certain medical conditions, such as bleeding disorders or allergies, may not be suitable for dermal fillers. Consider the benefits and risks of dermal fillers if your doctor says they’re an option for you. For dermal filler injections, the FDA recommends that you work with a licenced health care provider who has experience injecting dermal fillers as well as knowledge of fillers and anatomy, as well as an ability to manage complications.

What Are Dermal Fillers?
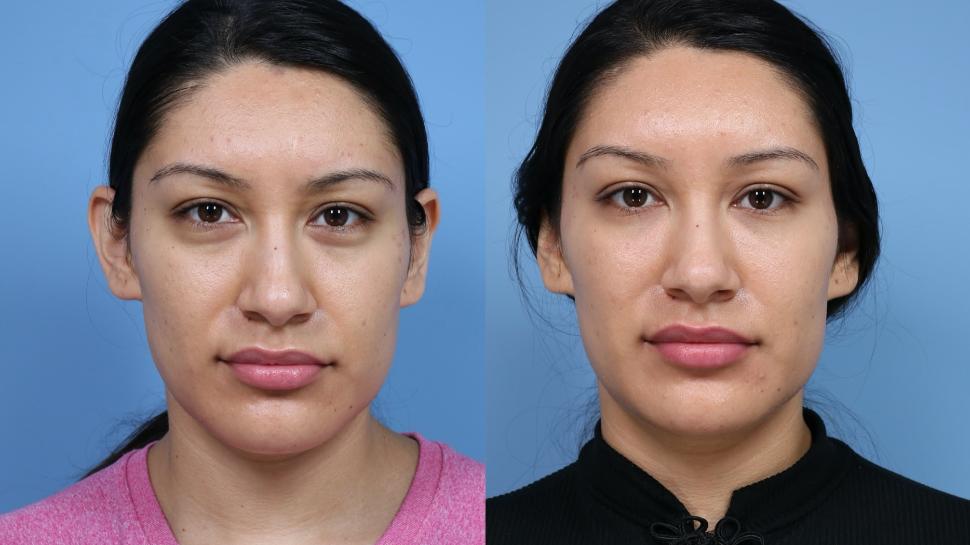
Dermal fillers are injectable gels that plump up hollow areas of the face. Dermal fillers can either smooth out wrinkles or plump up hollows in the face.
Dermal fillers are considered medical devices by the FDA. Most FDA-approved dermal fillers have short-term effects because they are made of materials that the body eventually breaks down and absorbs, according to clinical trials. Maintaining the desired effect may require a second injection.
Types of dermal fillers
Temporary fillers include the following materials:
- Hyaluronic acid, a sugar that is naturally found in the body
- Calcium hydroxylapatite, a mineral and a major component of bone
- Poly-L-lactic acid (PLLA), a biodegradable, synthetic material
A dermal filler that is not absorbed by the body is only FDA-approved. To make it, a solution containing bovine collagen and polymethylmethacrylate (PMMA) beads is mixed together. Plastic beads with smooth, round surfaces are known as PMMA beads.
Fda-approved Uses Of Dermal Fillers
Dermal fillers are approved for specific uses in people aged 22 and older. Those uses include:
- Correcting moderate-to-severe facial wrinkles and skin folds
- Increasing fullness of lips, cheeks, chin, under-eye hollows, jawline, and back of the hand
- Restoring facial fat loss in people with human immunodeficiency virus (HIV)
- Correcting acne scars on the cheek
Fda Warnings About Unapproved Fillers
- Silicone injections for body contouring and enhancement have not been cleared by the Food and Drug Administration (FDA). It’s not recommended to have filler injected into the breasts, buttocks, or between muscles. For large-scale body contouring or body enhancement, the injectable filler can result in long-term pain, infection, scarring or disfigurement, and even death.
- Injecting hyaluronic acid or other lip or facial fillers with needle-free devices is not recommended by the FDA, which has not approved them for use in injecting dermal fillers. It is difficult to control where the filler is injected because the injectors use a lot of pressure. Skin, lip, and eye damage can be severe and even permanent in some cases.
- Lip and facial fillers sold directly to the public should also be avoided, according to the FDA. They have not been approved by the FDA and may contain harmful chemicals or infectious organisms. FDA-approved dermal fillers must be administered by a trained medical professional using a cannula or a syringe with needles (a small flexible tubing with a blunt tip that is inserted under the skin).
Risks Of Fda-approved Fillers
Dermal fillers carry risks, just like any other medical procedure. As soon as an injection is given, most side effects are reported in clinical trials and post-market surveillance and quickly subside. This can take weeks, months or even years for side effects to show up.
Common risks include:
- Bruising
- Redness
- Swelling
- Pain
- Tenderness
- Itching
- Rash
- Difficulty in performing activities (only observed when injected into the back of the hand)
Before having dermal fillers made with certain materials, such as collagen, people should be tested for allergies.
Unintended injection into blood vessels
Dermal fillers carry the greatest risk of causing a blood vessel to burst. You may experience skin necrosis, stroke, or blindness if filler gets into your blood vessels. Despite the fact that the likelihood of this occurring is low, the consequences can be serious and even permanent if it does.
Removing Dermal Fillers
You may need additional procedures to reduce the filler’s amount or surgery to remove it if you want to reduce or remove it due to side effects. These procedures are not without their own set of dangers and hazards. Keep in mind that some filler materials may be difficult, if not impossible, to get rid of after the fact.
Also Read: Dave Chappelle Before And After: He Reveals Muscular New Look At NBA Finals
Dermal Fillers and Botulinum Toxin Products

Botulinum toxin products like Botox, Dysport, Xeomin, and Jeuveau have also been approved by the FDA to treat facial wrinkles. These are not fillers for the face. Because the muscles aren’t tense, the wrinkles don’t appear as prominently. Clinical studies have not examined the safety of using dermal fillers in conjunction with Botox and other treatments.
Despite the fact that botulinum toxin products are derived from the same bacteria that cause botulism, the amounts used for cosmetic purposes are purified and many orders of magnitude lower.
One or more types of facial lines, such as frown lines, forehead lines, and crow’s feet, have been approved by the FDA for temporary improvement in appearance.
Face weakness, eyelid drooping and drooping of the eyebrows have been reported in clinical trials. In addition, the injection site was swollen, reddened, and bruised, with other side effects such as localised pain. injections may cause dry eyes or difficulty swallowing or breathing in rare cases. While pregnant or lactating, it is not recommended to use botulinum toxin products for cosmetic purposes.

Irving is the Chief Editor at the Landscape Insight. He lives just outside of New York. His writings have also been featured in some very famous magazines. When he isn’t reading the source material for a piece or decompressing with a comfort horror movie, Irving is usually somewhere in his car. You can reach Irving at – [email protected] or on Our website Contact Us Page.








